Enclinical Trial Audit Plan Template - Request medical records to be reviewed Arrange for a quiet room Inform pharmacy of visit and schedule appointment Make sure PI and AIs will be available for monitoring date If multiple monitoring visits occur simultaneously make sure each sponsor has a separate room to ensure privacy and confidentiality
The clinical trial template has site lists of libraries for clinical trial protocols protocol documents announcements calendars issues tasks and document discussions These can be further customized with different versions of SharePoint To download this template you will need access to SharePoint Server 3 0
Enclinical Trial Audit Plan Template
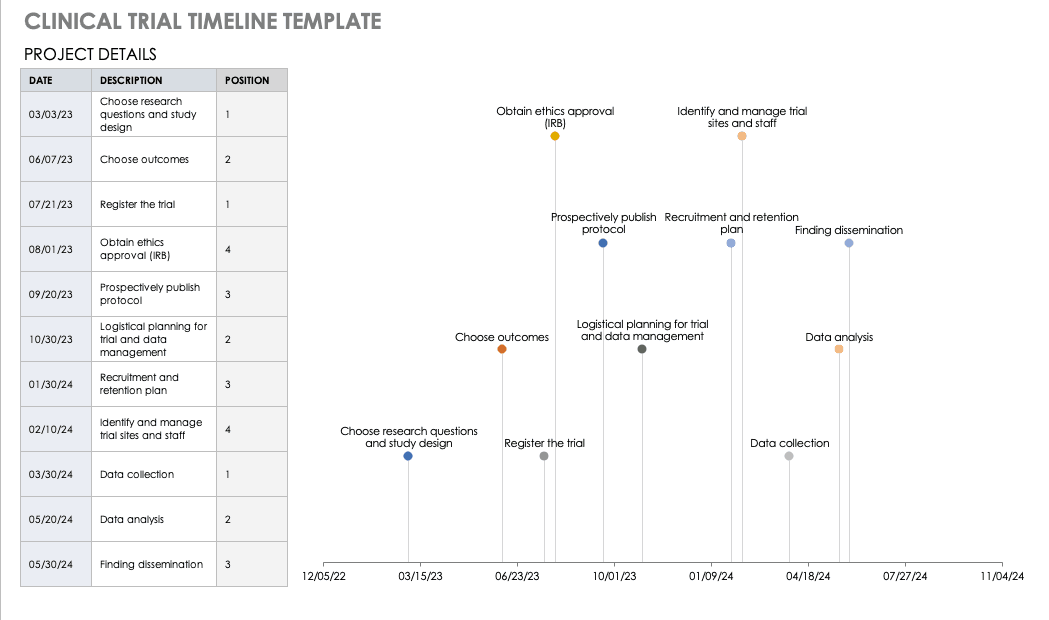
Enclinical Trial Audit Plan Template
Welcome to Global Health Trials' tools and templates library. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones have been added.
Audit plans such as an annual plan a monthly plan and a plan specific to each trial or audit should be established based on consideration of the goal s contents e g subjects and methods and timing of an audit the progress of the targeted trial and other relevant factors The audit plan should be updated in accordance with progress
Free Clinical Trial Templates Smartsheet
The PI and delegated research team members will prepare for the scheduled auditing visit by ensuring that all clinical research study related documents are current organized complete and accurate prior to the visit
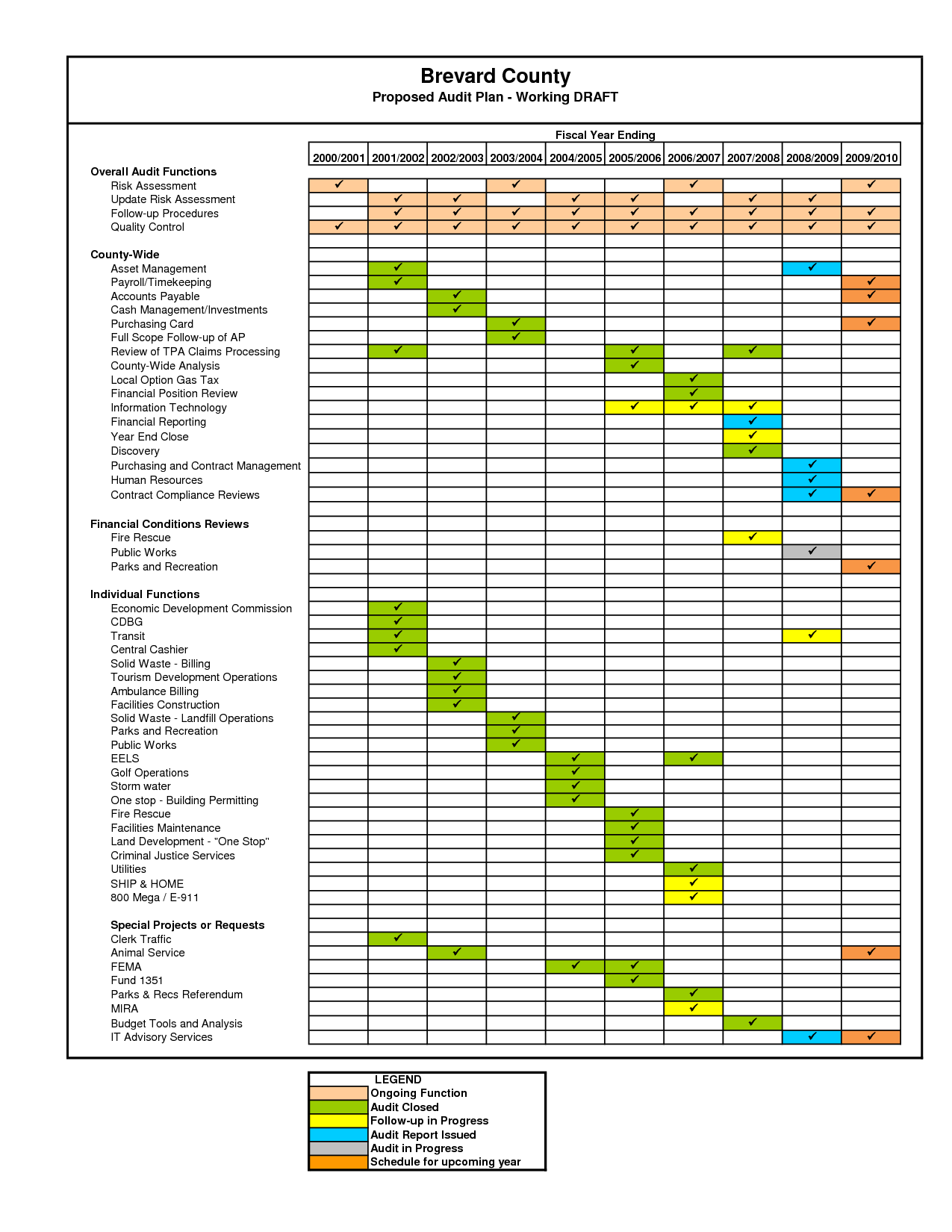
Internal Audit Plan Template Free FREE PRINTABLE TEMPLATES
American Society of Clinical Oncology Re search Community Forum Version September 2019 ASCO Toolkit FDA Audit Readiness 1 TOOLKIT OF RESOURCES FDA Audit Readiness
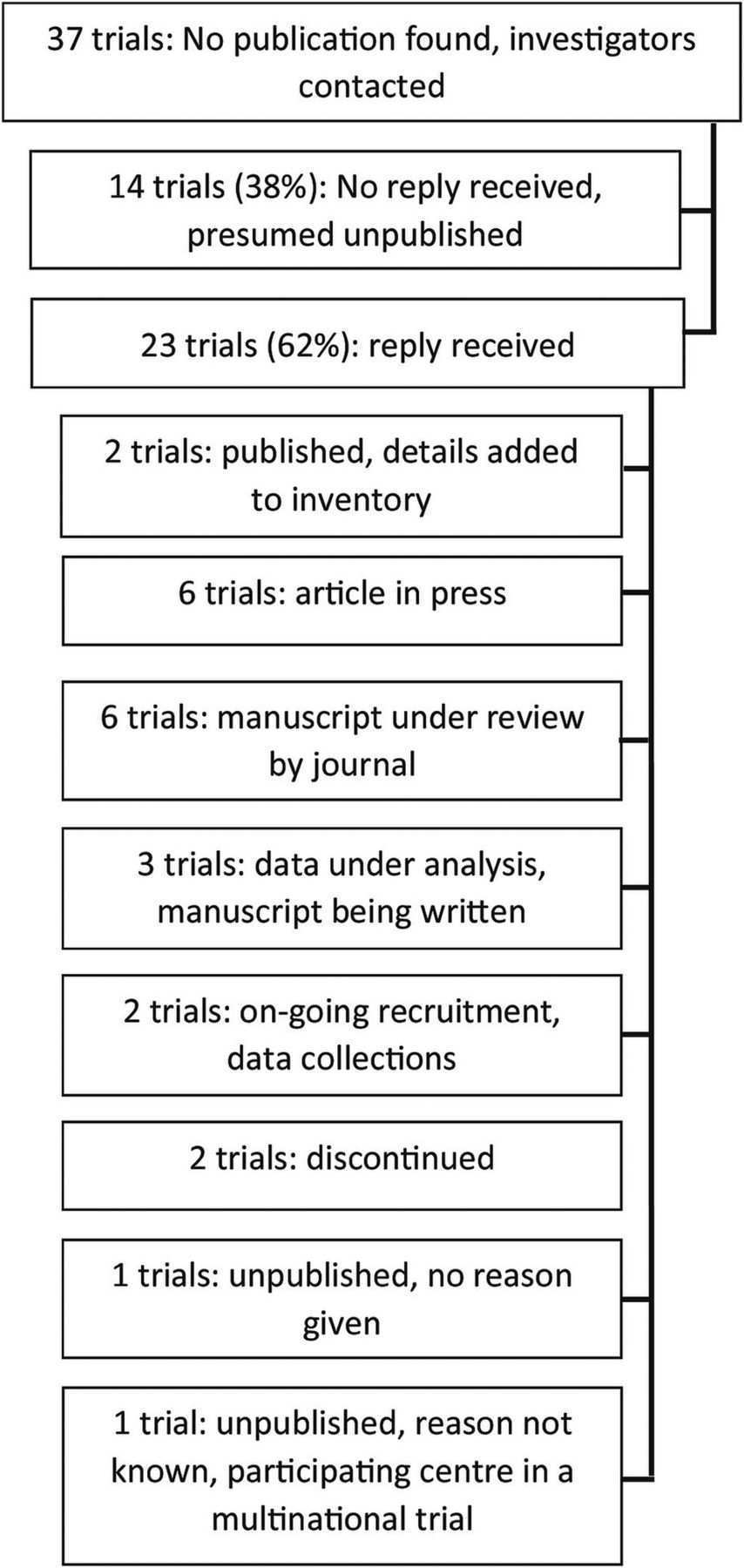
75 Printable Audit Plan Template For Clinical Trials Photo By Audit
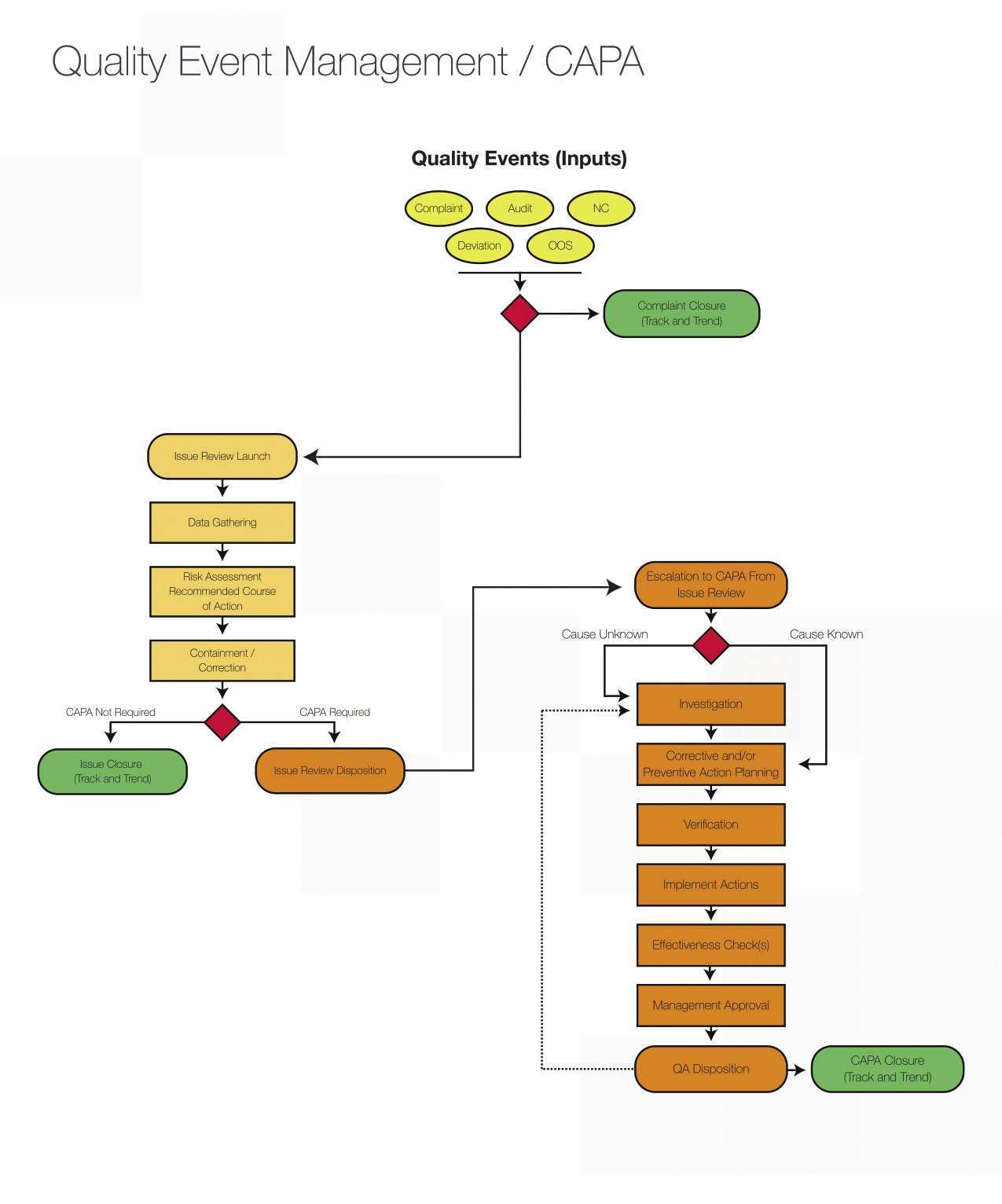
Audit Plan Template For Clinical Trials Cards Design Templates
Span Class Result Type
The Japan Society of Quality Assurance JSQA has prepared The JSQA GCP Guideline for GCP Auditing to promote the global discussion on GCP auditing and expects to establish a global guideline on GCP auditing along with ICH GCP The guideline will include the mission and the organization of a sponsor s auditing department and establish

Trial Report Template
AUDIT TRAIL REVIEW A KEY TOOL TO ENSURE DATA INTEGRITY An Industry Position Paper April 2021 Version PR1 Authors eClinical Forum eCF and the Society for Clinical Data Management SCDM
This course is intended to benefit regulatory good clinical practice (GCP) inspectors and sponsor auditors who audit clinical trials and in particular:
Downloadable Templates And Tools For Clinical Research
The Clinical Quality Assurance CQA audit program is a key element of a Good Clinical Practice GCP quality system The objective of a CQA audit program is to ensure that trial conduct and data quality meet protocol specified GCP and applicable regulatory requirements Due to the increasing complexity of clinical trials and regulatory
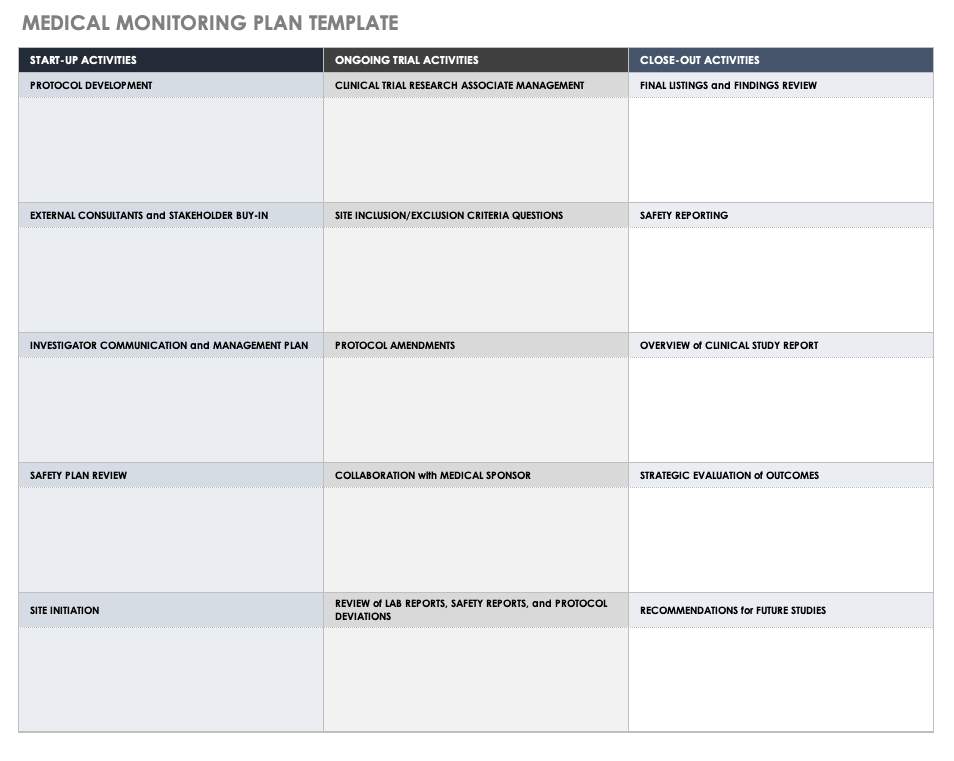
Audit Plan Template For Clinical Trials Cards Design Templates

Clinical Development Plan Template Fresh Spirit 2013 Explanation And
Enclinical Trial Audit Plan Template
AUDIT TRAIL REVIEW A KEY TOOL TO ENSURE DATA INTEGRITY An Industry Position Paper April 2021 Version PR1 Authors eClinical Forum eCF and the Society for Clinical Data Management SCDM
The clinical trial template has site lists of libraries for clinical trial protocols protocol documents announcements calendars issues tasks and document discussions These can be further customized with different versions of SharePoint To download this template you will need access to SharePoint Server 3 0

Pin On Report Template
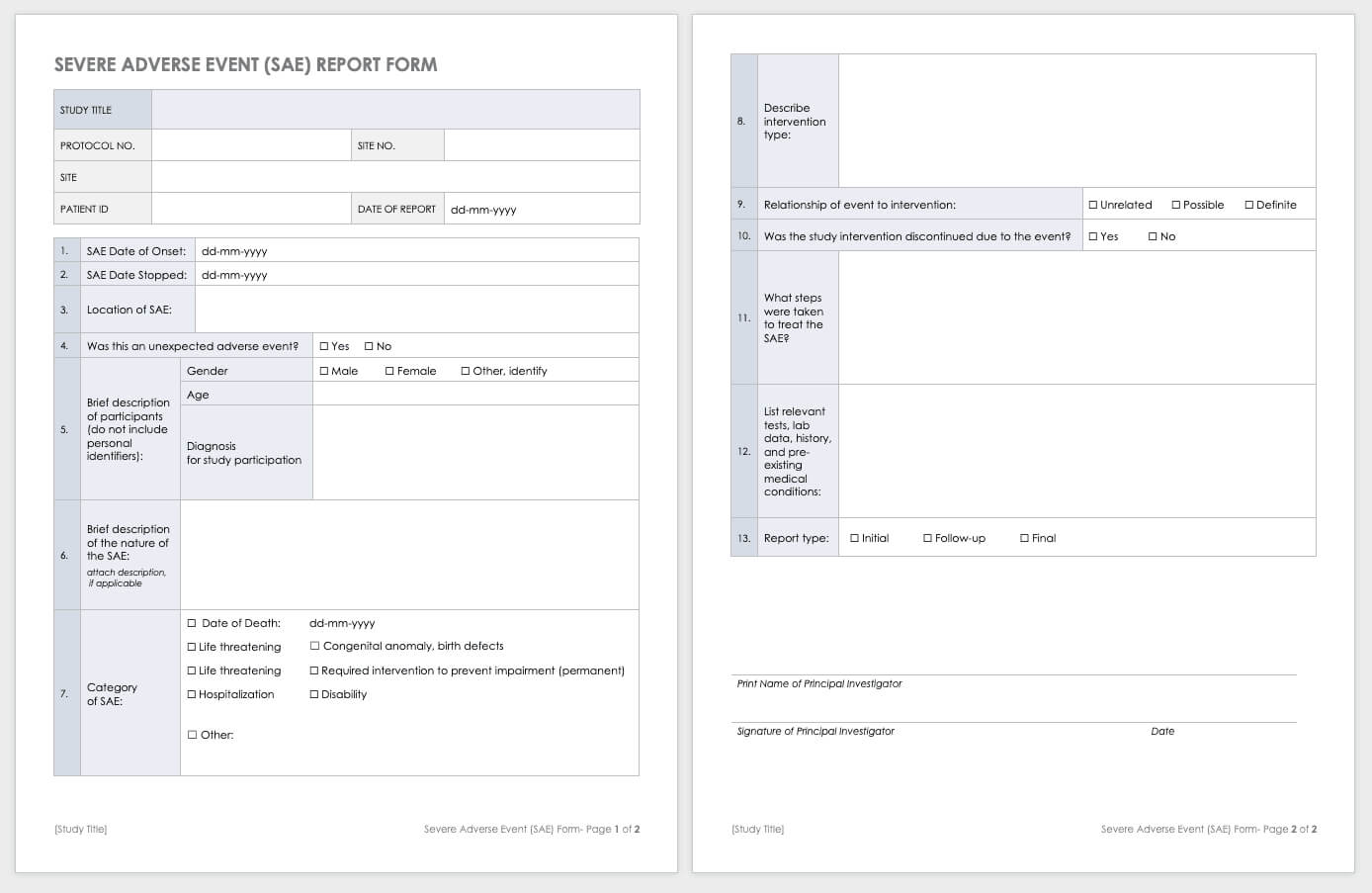
Free Clinical Trial Templates Smartsheet Inside Trial Report Template

The Enchanting The Basics Of Clinical Trial Centralized Monitoring For

Free Clinical Trial Templates Smartsheet In Trial Report Template

Case Report Form Template