Ennaltrexone Medication Template - Naltrexone has a stronger pull to the brain s opioid receptor This means that when it enters the brain it will knock out other opioids such as heroin that are sitting in the brain s opioid receptor This causes a person to rapidly enter withdrawal Normally withdrawal comes on gradually as opioids slowly lose their effect
Naltrexone also modifies how the hypothalamus pituitary gland and adrenal gland hypothalamic pituitary adrenal axis HPA axis interact to suppress the amount of alcohol consumed Naltrexone comes as an extended release intramuscular injection Vivitrol and as oral tablets The branded versions of naltrexone tablets Revia Depade have
Ennaltrexone Medication Template

Ennaltrexone Medication Template
Naltrexone is an FDA-approved opioid antagonist used to treat alcohol use disorder and opioid dependence. Naltrexone blocks the effect of opioids and prevents opioid intoxication and physiologic dependence on opioid users. This medication is a mu-opioid receptor antagonist and also a weaker antagonist of the kappa and delta-opioid receptors. This activity is designed to improve the knowledge ...
Medicine to treat a cold cough diarrhea or pain in 7 14 ausnea vomiting loss of appetite joint pain muscle cramps dizziness drowsiness sleep problems insomnia tooth pain or cold symptoms such as stuffy nose sneezing sore throat Blocks the effects of opioids in previously
Naltrexone Uses Dosage Side Effects Warnings Drugs
Step 4 Assessement Patient Readiness Initial readiness assessment for XR naltrexone 1 Vital signs 2 Urine toxicology screen for all opioids including buprenorphine

Nursing Medication Template Etsy
Trouble sleeping Feeling dizzy sleepy tired or weak Back muscle or joint pain Signs of a common cold Tooth pain Injection Irritation where the shot is given

Medication YouTube

481 Week 5 Med PDF Medication Template MEDICATION WORKSHEET Azalea
Span Class Result Type
Administered every 4 hours to attenuate the withdrawal induced by naltrexone highest mean dosage was 2 3 mg daily on day 3 Administered every 4 hours dosage increased in 2 mg increments during the daytime on day 3 Day 4 Administered only as needed to reduce signs and symptoms of withdrawal 10 mg 3 times daily

Medication Handwriting Image
The FDA requires warnings on formulations of naltrexone about possible liver adverse effects The current product labeling for extended release injectable naltrexone
ATI Templates active learning template: system disorder student caring for client who is recoving from alcohol use disorder 12
Naltrexone StatPearls NCBI Bookshelf
M EDICATION F ACT S HEET N ALTREXONE 4 Summary of Black Box Warnings Naltrexone does not have any black box warnings Important Disclosure This information is being provided as a community outreach effort of the American Association of Psychiatric Pharmacists

System Detects Errors When Medication Is Self administered MIT News

Blank Medication Allbusinesstemplates
Ennaltrexone Medication Template
The FDA requires warnings on formulations of naltrexone about possible liver adverse effects The current product labeling for extended release injectable naltrexone
Naltrexone also modifies how the hypothalamus pituitary gland and adrenal gland hypothalamic pituitary adrenal axis HPA axis interact to suppress the amount of alcohol consumed Naltrexone comes as an extended release intramuscular injection Vivitrol and as oral tablets The branded versions of naltrexone tablets Revia Depade have

12 Medication Worksheet Printable Worksheeto

Adderall ADHD Medication With A Composition Intended To Av Flickr
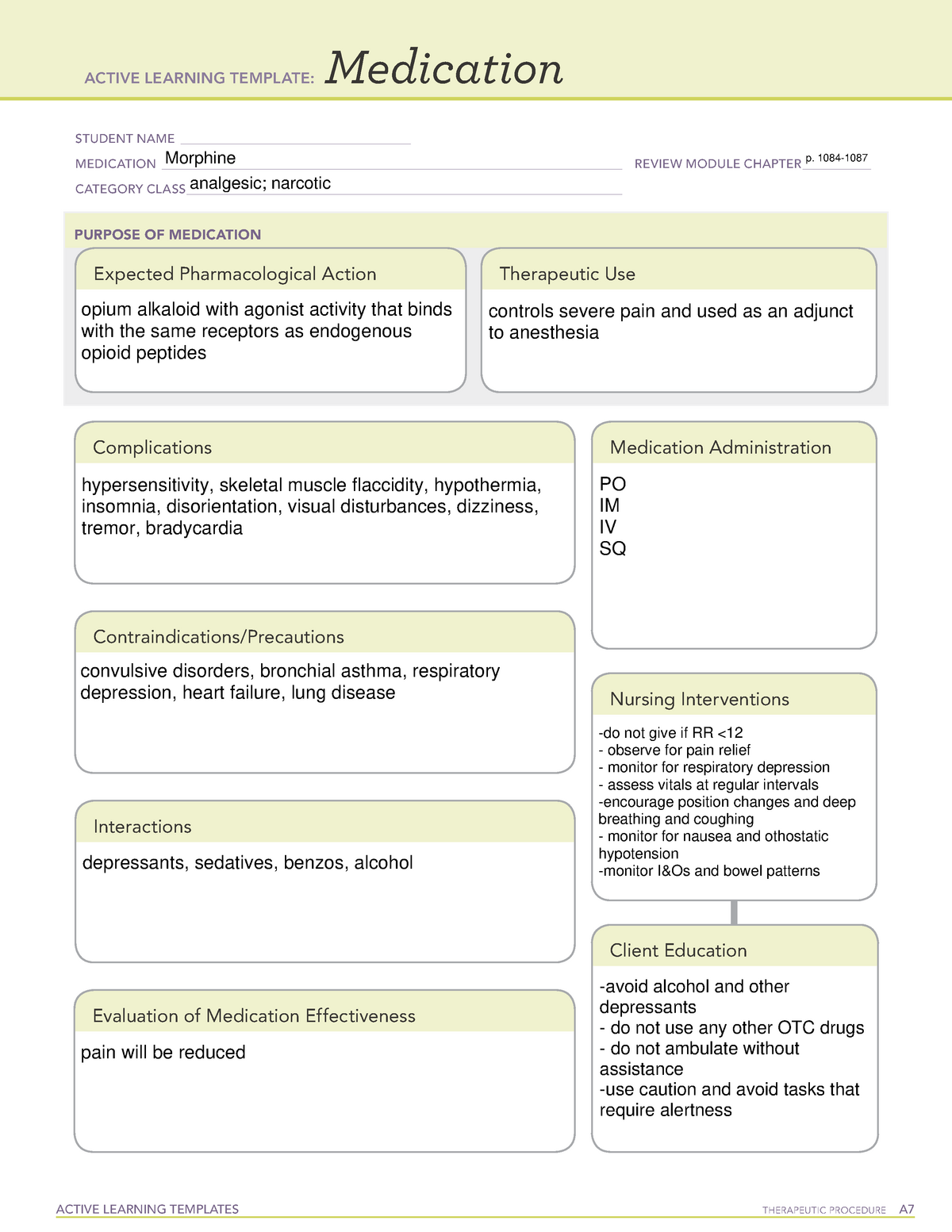
Enoxaparin Ati Medication Template

Medication Administration Record Printable

Medication Administration YouTube