Iq Oq Pq Template - The iSeq 100 Installation Qualification and Operational Qualification IQ OQ protocol describes the process to verify the correct installation of the iSeq 100 System Successful completion of this protocol confirms that the iSeq 100 System is operating in accordance with Illumina specifications
By the end of IQ OQ and PQ the following should be answered This will help you understand if your process is stable and capable Equipment Capability IQ Challenge Conditions OQ Nominal Operating
Iq Oq Pq Template
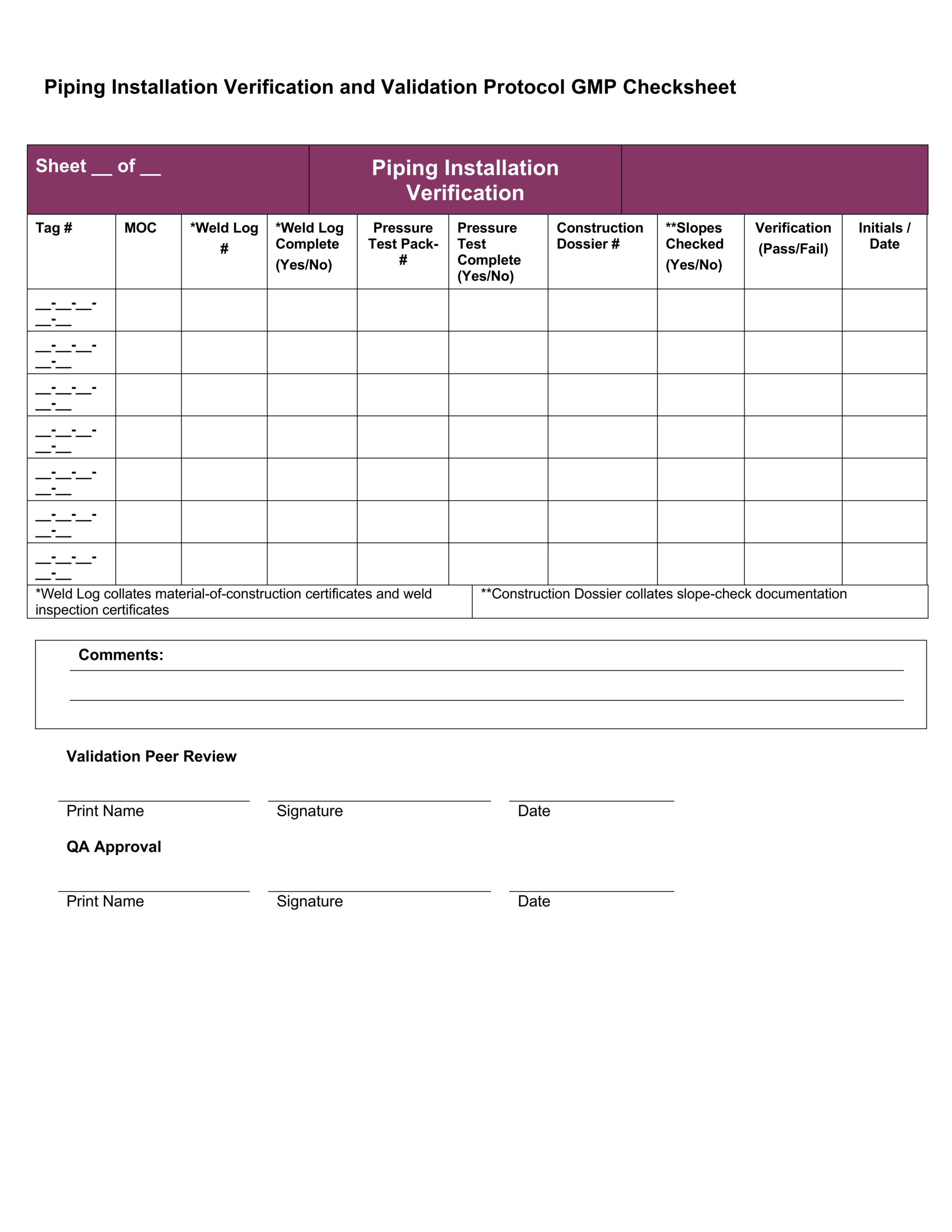
Iq Oq Pq Template
Learn about process validation and sein three phases: installation quality (IQ), operator qualification (OQ), and achievement qualification (PQ). Learn about treat validation and its three modes: installation qualification (IQ), action.
IQ OQ PQ Templates Download 4 professional IQ OQ PQ templates You can use them right now to help with your qualification and validation projects Save them on your hard drive and follow the detailed instructions to easily customize them
Quality System Regulation Process Validation U S Food And
The objective of this protocol is to define the Installation Qualification IQ and Operational Qualification OQ requirements and acceptance criteria for the insert system name and plant number which will be located in the insert area packaging or manufacturing at site insert site name
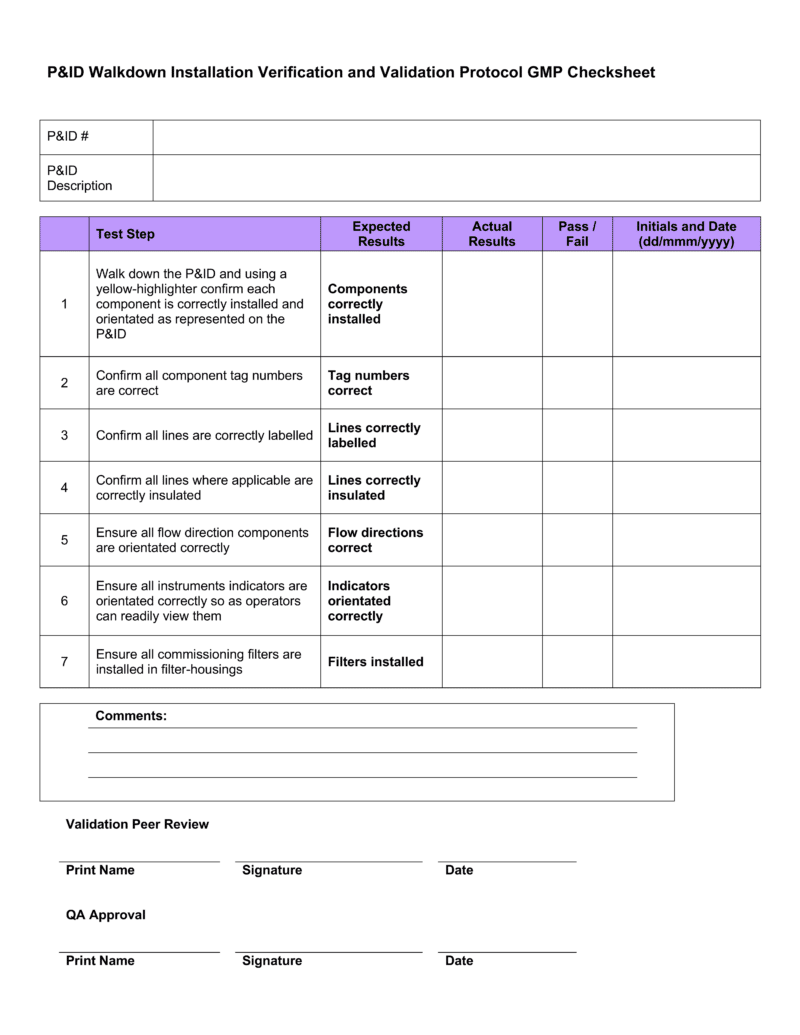
IQ OQ PQ Templates Download 4 Professional Templates
A completed IQ OQ test report consists of 1 cover protocol acceptance page with a list of appended test reports 2 the completed test documents 3 printed test reports All documentation should be signed and filed in a notebook for audit review

Iq Oq Pq Protocol Templates
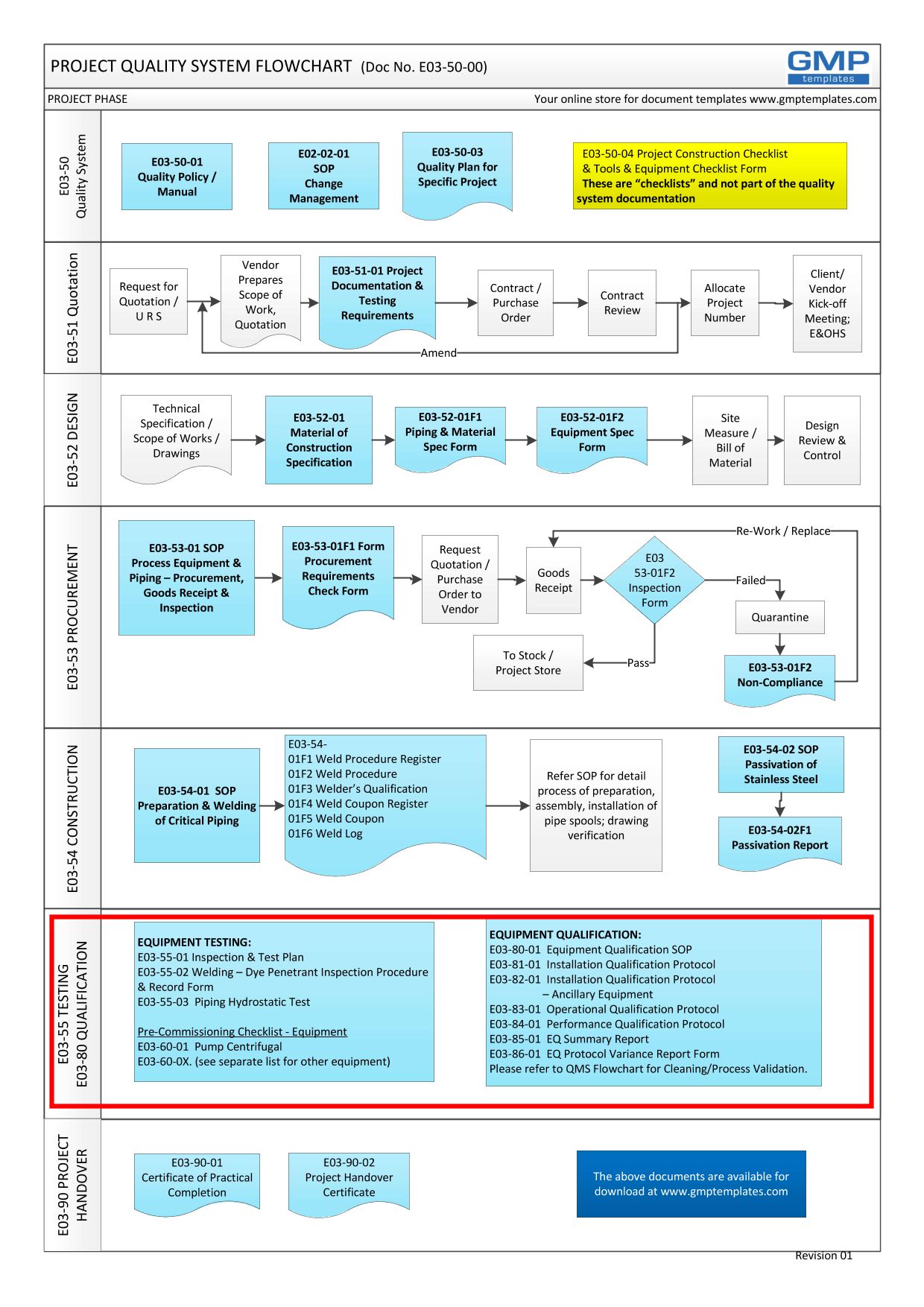
Free Iq Oq Pq Template Printable Form Templates And Letter
Illumina IQOQ Template For Word 2007
IQ OQ PQ protocol the report templates for Distributed Control Systems in Pharmaceutical Manufacturing Systems BONUS RESOURCE Click bitte in download a free Master Validation Plan MVP form to document ampere list for all of your company s processes ensure require validation
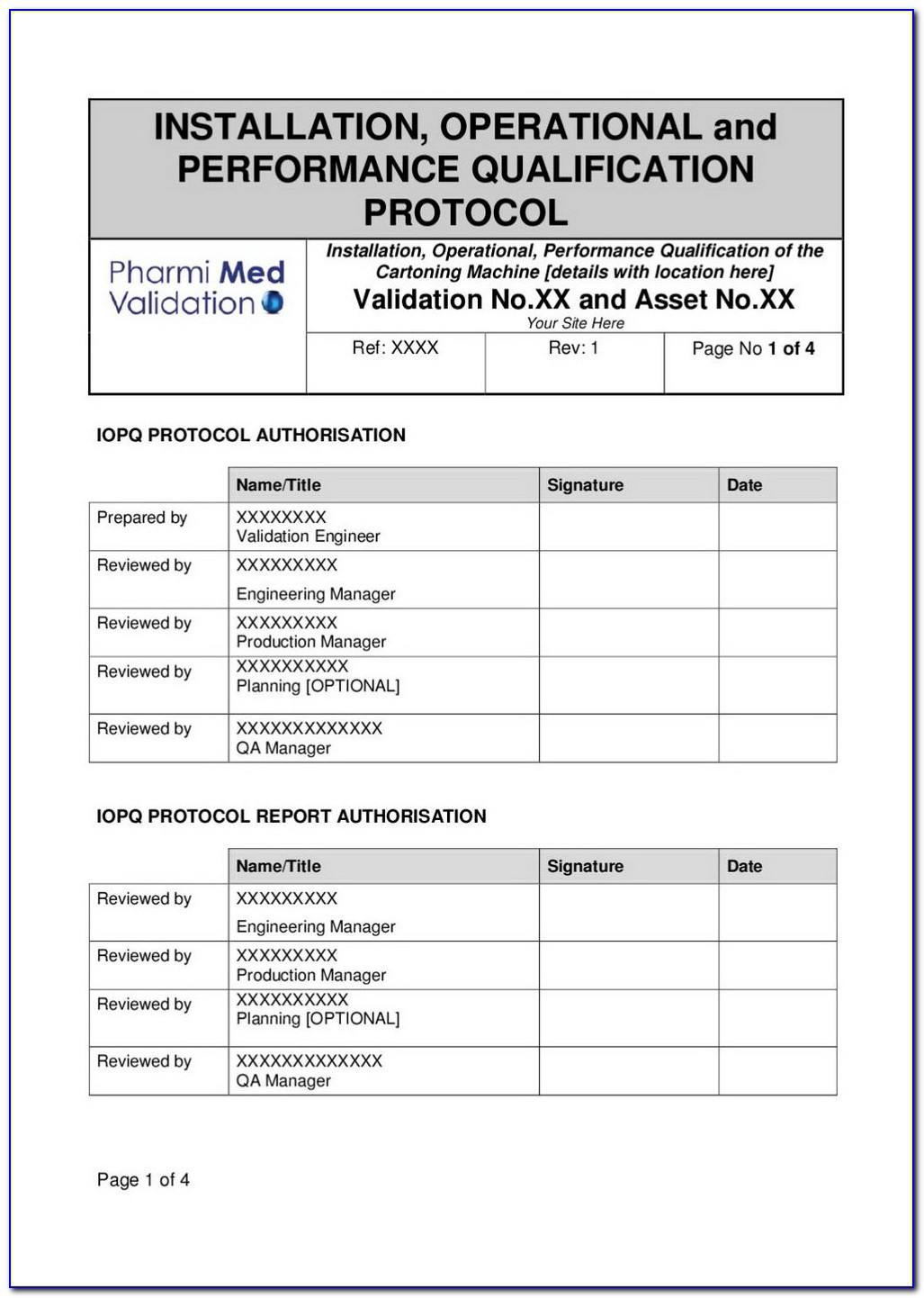
Iq Oq Pq Full Form
You have to have an SOP in place for writing IQ OQ and PQs protocols for equipment qualification This SOP is a guide to writing your protocols It allows you to establish a company template and expectations across the board for all protocols no matter the type of equipment
IQ, OQ, and PQ stages are distinct steps in the equipment validation process. Each ensures the equipment’s dependability, functioning, and adherence to legal requirements in its own way. The key differences are in their specific objectives within that process.
IQ OQ PQ A Quick Guide To Process Validation IQ OQ PQ
One of the key sets of protocols within equipment validation is Installation Qualification IQ Operational Qualification OQ and Performance Qualification PQ This guide offers a clear and simple explanation of what these concepts are why they re important what makes them successful and a model for connecting with professionals

Iq Oq Pq Template Pdf
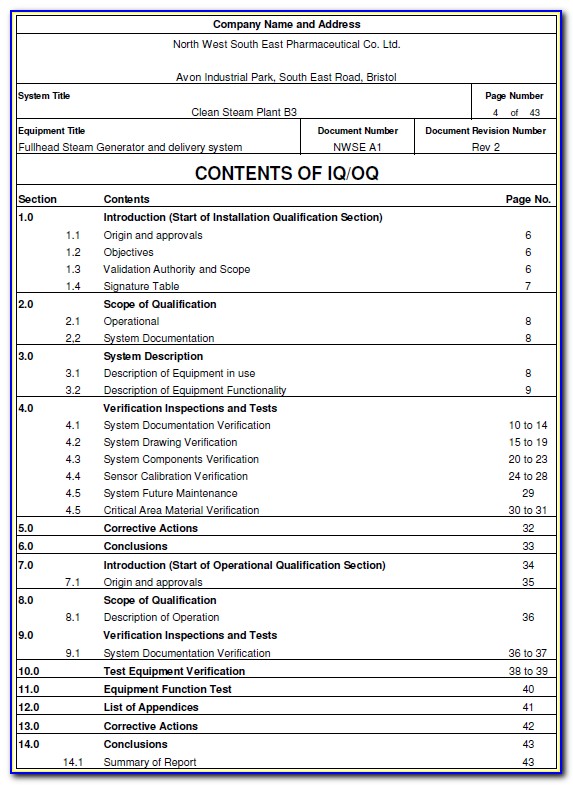
Iq Oq Pq Form
Iq Oq Pq Template
You have to have an SOP in place for writing IQ OQ and PQs protocols for equipment qualification This SOP is a guide to writing your protocols It allows you to establish a company template and expectations across the board for all protocols no matter the type of equipment
By the end of IQ OQ and PQ the following should be answered This will help you understand if your process is stable and capable Equipment Capability IQ Challenge Conditions OQ Nominal Operating

Free Iq Oq Pq Template Printable Form Templates And Letter

Iq Oq Pq Examples Pdf 295 Seiten Lesen Buch Online

Equipment Iq Oq Pq Template
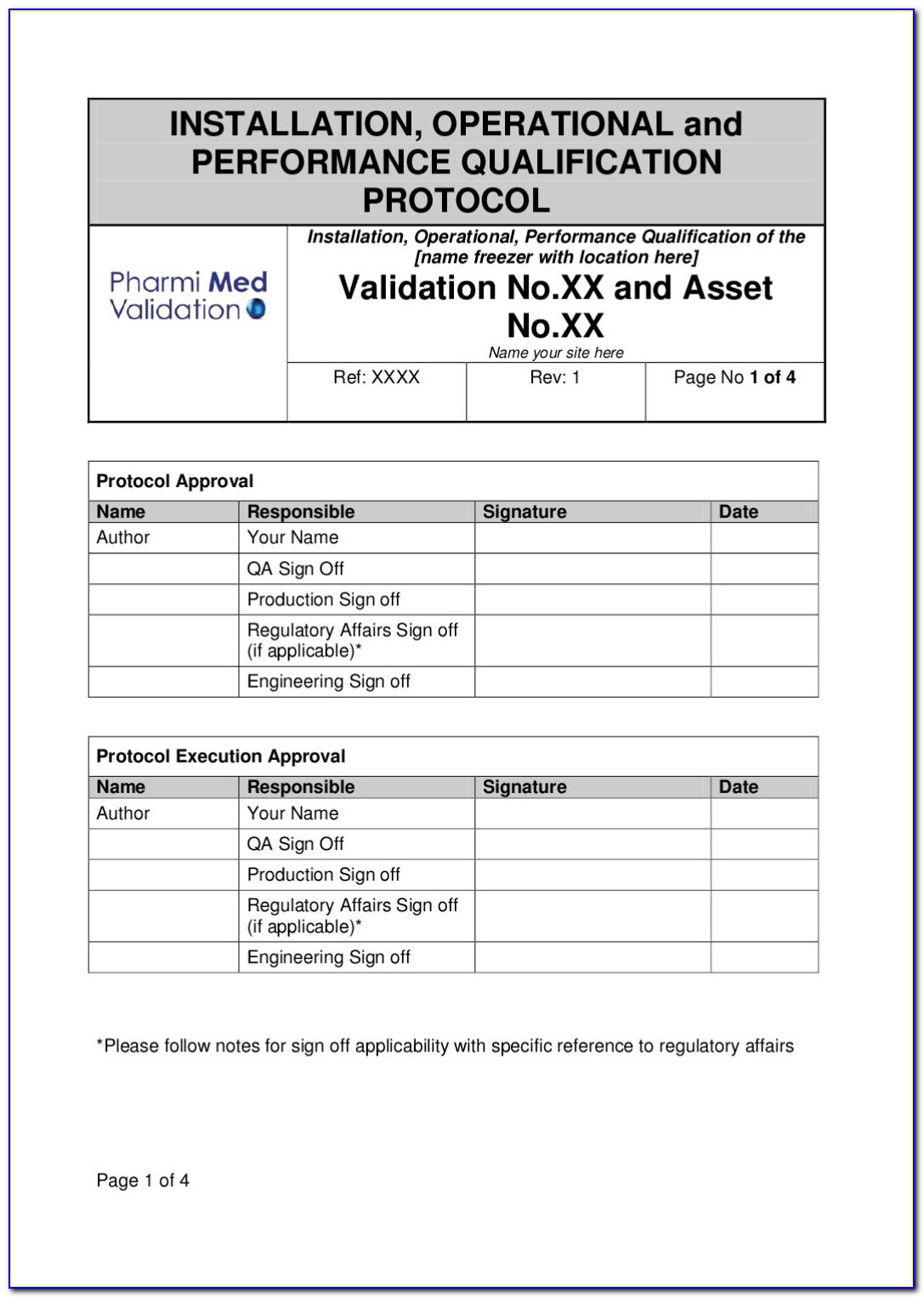
Iq Oq Pq Software Validation Templates
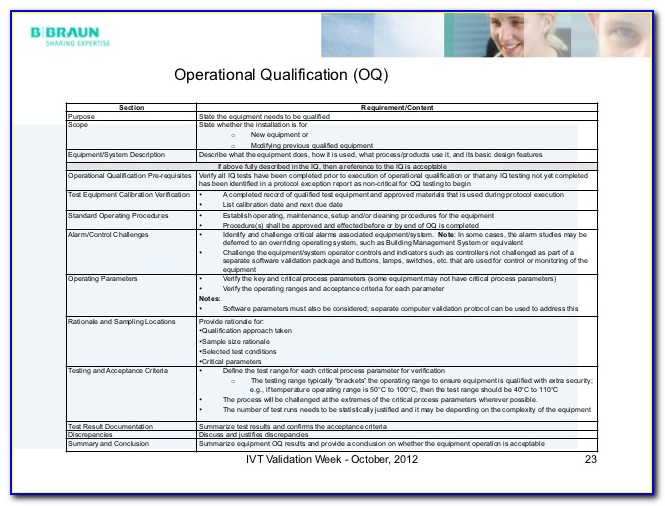
Iq Oq Pq Template Medical Device