Sculptra Consent Form PDF - SCULPTRA Aesthetic is indicated for use in immune competent people as a single regimen for correction of shallow to deep nasolabial fold contour deficiencies and other facial wrinkles in which deep dermal grid pattern cross hatch injection technique is appropriate
Sculptra Aesthetic is an injectable implant that contains microparticles of L Poly Lactic Acid a biocompatible biodegradable synthetic polymer from the alpha hydroxy acid family A treatment regimen of SCULPTRA consists of up to 4 injection sessions scheduled 3 4 weeks apart
Sculptra Consent Form PDF

Sculptra Consent Form PDF
Sculptra takes time to gradually correct the depression in your skin. Your doctor will decide the appropriate number of treatment sessions and the amount of Sculptra you will need at each session. Multiple sessions are often required and patients with severe facial fat loss may require 3 to 6 treatment sessions.
INFORMED CONSENT FORM Sculptra is a sterile suspension of Poly L Lactic acid which is a biocompatible does not harm the body synthetic polymer from the alpha hydroxy acid family fruit acids Poly L Lactic acid has been used medically for many years in dissolvable stitches and does not require pre treatment skin testing for allergies
Span Class Result Type
INFORMED CONSENT FOR SCULPTRA This document will inform you about your Sculptra Injectable procedure the risks associated with this procedure and alternative treatments It is important that you read this information carefully and completely After reviewing please sign the consent authorizing the procedure to be performed

Galderma Sculptra Consent Form Fill Online Printable Fillable Blank PdfFiller
Sculptra therapy is designed to help correct skin depression such as creases wrinkles folds scars hollow eye rings degenerative skin aging and facial lipotrophy loss of fat Sculptra is a poly L lactic implant in the form of a sterile apyrogenic suspension

UK Sculptra Consent Form

Sculptra Consent Forms Editable Aesthetics Forms Dermal Etsy
Span Class Result Type
This is an informed consent document which has been prepared to help your plastic surgeon inform you concerning Radiesse and Sculptra injections and its risks It is important that you read this information carefully and completely

Sculptra Consent Form
INFORMED CONSENT SCULPTRA AESTHETIC TREATMENT Rev 5 23 Overview Sculptra Aesthetic is an FDA approved injectable poly L lactic acid implant Poly L lactic acid is a biocompatible biodegradable synthetic polymer
Consent Form for Sculptra Treatment Page 1 of 3 Consent Form for Sculptra Treatment This is a patient consent form for treatment using Sculptra. Please read it carefully before signing. This disclosure is not meant to alarm you; it is simply an effort to better inform you. Being informed will help you make the decision whether to undergo treatment.
Span Class Result Type
SCULPTRA is a sterile injectable biocompatible biodegradable material that is made of very small particles of a synthetic polymer named poly L lactic acid PLLA carboxymethylcellulose USP non pyrogenic mannitol USP and sterile water for injection USP

Sculptra Consent Form Sculptra Aesthetic Injection Consent Etsy

Consent Form Clinical Research Glossary
Sculptra Consent Form PDF
INFORMED CONSENT SCULPTRA AESTHETIC TREATMENT Rev 5 23 Overview Sculptra Aesthetic is an FDA approved injectable poly L lactic acid implant Poly L lactic acid is a biocompatible biodegradable synthetic polymer
Sculptra Aesthetic is an injectable implant that contains microparticles of L Poly Lactic Acid a biocompatible biodegradable synthetic polymer from the alpha hydroxy acid family A treatment regimen of SCULPTRA consists of up to 4 injection sessions scheduled 3 4 weeks apart
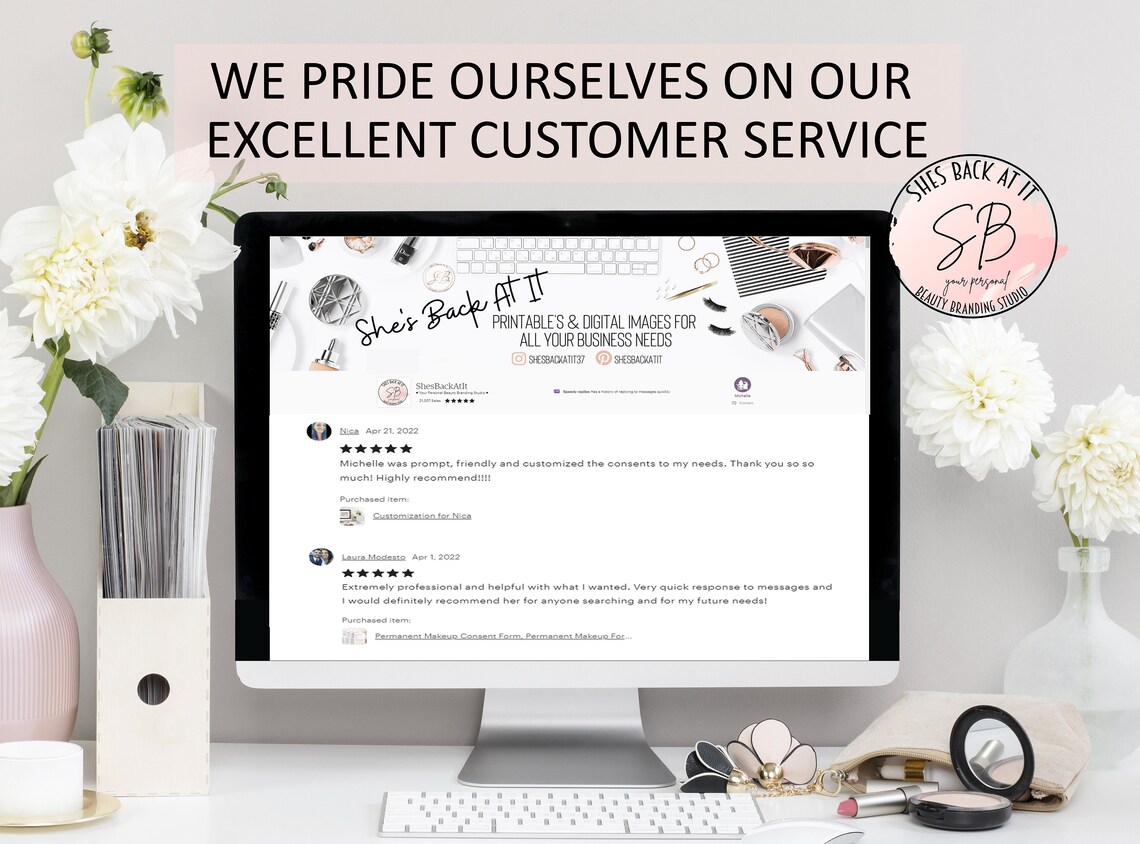
Sculptra Consent Form Sculptra Aesthetic Injection Consent Etsy Australia

Sculptra Consent Form Sculptra Aesthetic Injection Consent Etsy Finland

Sculptra Consent First Glance Aesthetic Clinic

Sculptra Filler Informed Consent Form Template Pack Nurse Etsy

Sculptra Consent Form Sculptra Aesthetic Injection Consent Etsy Finland